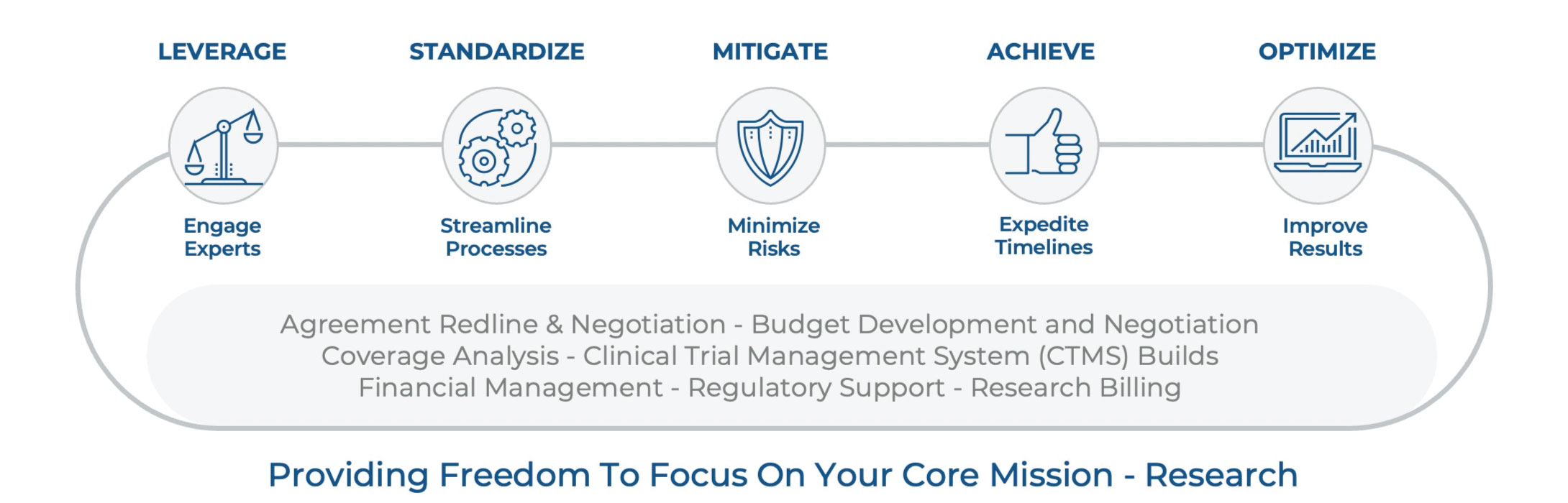
Full lifecycle clinical trials support
Clinical trials have led to some of the most important breakthroughs in research. Increase the pace of discovery by collaborating with Huron to reduce risk, effectively manage costs, ensure compliance, and achieve success throughout the research life cycle.
How we help